| In vitro: |
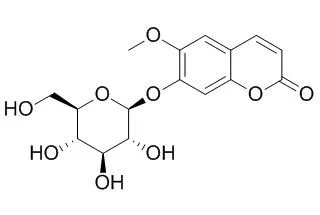
| BMC Plant Biol . 2014 Oct 18;14:280. | | Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana[Pubmed: 25326030] | | Abstract
Background: Scopoletin and its glucoside Scopolin are important secondary metabolites synthesized in plants as a defense mechanism against various environmental stresses. They belong to coumarins, a class of phytochemicals with significant biological activities that is widely used in medical application and cosmetics industry. Although numerous studies showed that a variety of coumarins occurs naturally in several plant species, the details of coumarins biosynthesis and its regulation is not well understood. It was shown previously that coumarins (predominantly Scopolin and scopoletin) occur in Arabidopsis thaliana (Arabidopsis) roots, but until now nothing is known about natural variation of their accumulation in this model plant. Therefore, the genetic architecture of coumarins biosynthesis in Arabidopsis has not been studied before.
Results: Here, the variation in Scopolin and scopoletin content was assessed by comparing seven Arabidopsis accessions. Subsequently, a quantitative trait locus (QTL) mapping was performed with an Advanced Intercross Recombinant Inbred Lines (AI-RILs) mapping population EstC (Est-1 × Col). In order to reveal the genetic basis of both Scopolin and scopoletin biosynthesis, two sets of methanol extracts were made from Arabidopsis roots and one set was additionally subjected to enzymatic hydrolysis prior to quantification done by high-performance liquid chromatography (HPLC). We identified one QTL for Scopolin and five QTLs for scopoletin accumulation. The identified QTLs explained 13.86% and 37.60% of the observed phenotypic variation in Scopolin and scopoletin content, respectively. In silico analysis of genes located in the associated QTL intervals identified a number of possible candidate genes involved in coumarins biosynthesis.
Conclusions: Together, our results demonstrate for the first time that Arabidopsis is an excellent model for studying the genetic and molecular basis of natural variation in coumarins biosynthesis in plants. It additionally provides a basis for fine mapping and cloning of the genes involved in Scopolin and scopoletin biosynthesis. Importantly, we have identified new loci for this biosynthetic process. |
|
| In vivo: |
| Int Immunopharmacol. 2009 Jul;9(7-8):859-69. | | Scopolin isolated from Erycibe obtusifolia Benth stems suppresses adjuvant-induced rat arthritis by inhibiting inflammation and angiogenesis.[Pubmed: 19327410] | Despite Scopolin is a main coumarin constituent in the stems of Erycibe obtusifolia Benth, a herb drug that has long been utilized in traditional Chinese medicine for the treatment of rheumatoid arthritis, little information is available about the pharmacological activities of this compound.
The present study was performed to investigate the anti-rheumatic effects of Scopolin in adjuvant-induced arthritis (AIA) in rats, and explore the underlying mechanisms of action in views of anti-inflammatory and anti-angiogenic properties in the synovial tissues.
METHODS AND RESULTS:
Scopolin (50, 100 mg/kg), injected intraperitoneally for 10 days from the onset of secondary response, significantly inhibited both inoculated and non-inoculated paw swelling as well as articular index scores in AIA. Meanwhile, the mean body weight of rats treated with Scopolin was higher than that of model group. Rats treated with high dose of Scopolin (100 mg/kg) preserved a nearly normal histological architecture of the joints and showed a significant reduction of the new blood vessels in the synovial tissues. Additionally, Scopolin could reduce IL-6, VEGF and FGF-2 expressions in rat synovial tissues.
CONCLUSIONS:
In conclusion, Scopolin can reduce the clinical symptoms of rat AIA by inhibiting inflammation and angiogenesis, and this compound may be a potent agent for angiogenesis related diseases and can serve as a structural base for screening more potent synthetic analogs. | | Euphytica, 2006, 147(3):451-460. | | Fungitoxic effect of scopolin and related coumarins on Sclerotinia sclerotiorum. A way to overcome sunflower head rot.[Reference: WebLink] | The content of coumarins, as probable phytoalexins, was analysed in four sunflower genotypes that ranged in responses to head rot from highly susceptible to highly resistant.
METHODS AND RESULTS:
Low levels of all coumarins (Scopolin, scopoletin and ayapin) were detected in the three most susceptible genotypes irrespective of time after inoculation. However, in the resistant genotype there was a clear time-dependent disease-induced increase of all coumarins that reached a maximum after 10–14 days. Detailed comparison of the most susceptible and the resistant genotype showed that in the resistant but not the susceptible, scopoletin peroxidase activity increased during the course of the experiment. Results confirmed a clear negative correlation between coumarin content and disease symptoms and in particular for Scopolin. Furthermore we show for the first time that Scopolin is inhibitory to Sclerotinia at similar doses to scopoletin.
CONCLUSIONS:
As Scopolin is known to be less phytotoxic than ayapin and scopoletin, its accumulation may well confer head rot resistance with minimal plant damage and might be one of the bases for resistance to Sclerotinia. | | Sci Rep . 2017 May 22;7(1):2251. | | Scopolin ameliorates high-fat diet induced hepatic steatosis in mice: potential involvement of SIRT1-mediated signaling cascades in the liver[Pubmed: 28533555] | | Abstract
The present study aimed to investigate whether Scopolin exhibits beneficial effects on high-fat diet (HFD)-induced hepatic steatosis in mice. The involvement of sirtuin 1 (SIRT1) as a molecular target for Scopolin was also explored. Scopolin decreased the Km of SIRT1 for p53 and nicotinamide adenine dinucleotide without altering Vmax in a cell-free system. Scopolin alleviated oleic acid-induced lipid accumulation and downregulation of SIRT1 activity in HepG2 cells, and these beneficial effects of Scopolin were abolished in the presence of SIRT1 inhibitor. Mice administered 0.02% Scopolin for 8 weeks exhibited improved phenotypes of HFD-induced hepatic steatosis along with increased hepatic SIRT1 activity and protein expression. Scopolin resulted in increased deacetylation of sterol regulatory element-binding protein 1c with subsequent downregulation of lipogenic genes, and enhanced deacetylation of protein peroxisome proliferator-activated receptor-γ coactivator 1α with upregulation of fatty acid oxidation genes in livers. Scopolin also enhanced deacetylation of nuclear factor-kappa enhancer binding protein and liver kinase B1 (LKB1), facilitating LKB1/AMP-activated protein kinase signaling cascades. Scopolin attenuated hepatic steatosis through activation of SIRT1-mediated signaling cascades, a potent regulator of lipid homeostasis. Increased hepatic SIRT1 activity and protein expression appeared to be associated with these beneficial effects of Scopolin. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)