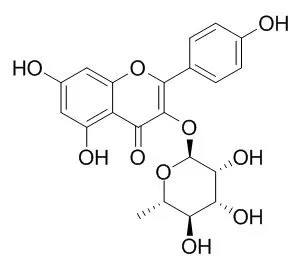
| Description: |
Afzelin has several cellular activities such as DNA-protective, antibacterial, antioxidant, and anti-inflammatory as well as UV-absorbing activity and may protect human skin from UVB-induced damage by a combination of UV-absorbing and cellular activities.
Afzelin has potenial anti-cancer activity against prostate cancer, the activity is due to inhibition of LIM domain kinase 021 expression, it can inhibit the proliferation of LNCaP and PC302cells, and block the cell cycle in the G002phase. Afzelin can attenuate asthma phenotypes is based on reduction of Th2 cytokine via inhibition of GATA-binding protein 3 transcription factor, which is the master regulator of Th2 cytokine differentiation and production. |
| In vitro: |
| PLoS One. 2013 Apr 23;8(4):e61971. | | Antagonizing effects and mechanisms of afzelin against UVB-induced cell damage.[Pubmed: 23626759] | Ultraviolet (UV) radiation induces DNA damage, oxidative stress, and inflammatory processes in human keratinocytes, resulting in skin inflammation, photoaging, and photocarcinogenesis. Adequate protection of skin against the harmful effects of UV irradiation is essential.
METHODS AND RESULTS:
Therefore, in this study, we investigated the protective effects of Afzelin, one of the flavonoids, against UV irradiation in human keratinocytes and epidermal equivalent models. Spectrophotometric measurements revealed that the Afzelin extinction maxima were in the UVB and UVA range, and UV transmission below 376 nm was <10%, indicating UV-absorbing activity of Afzelin. In the phototoxicity assay using the 3T3 NRU phototoxicity test (3T3-NRU-PT), Afzelin presented a tendency to no phototoxic potential. In addition, in order to investigate cellular functions of Afzelin itself, cells were treated with Afzelin after UVB irradiation. In human keratinocyte, Afzelin effectively inhibited the UVB-mediated increase in lipid peroxidation and the formation of cyclobutane pyrimidine dimers. Afzelin also inhibited UVB-induced cell death in human keratinocytes by inhibiting intrinsic apoptotic signaling. Furthermore, Afzelin showed inhibitory effects on UVB-induced release of pro-inflammatory mediators such as interleukin-6, tumor necrosis factor-α, and prostaglandin-E2 in human keratinocytes by interfering with the p38 kinase pathway. Using an epidermal equivalent model exposed to UVB radiation, anti-apoptotic activity of Afzelin was also confirmed together with a photoprotective effect at the morphological level.
CONCLUSIONS:
Taken together, our results suggest that Afzelin has several cellular activities such as DNA-protective, antioxidant, and anti-inflammatory as well as UV-absorbing activity and may protect human skin from UVB-induced damage by a combination of UV-absorbing and cellular activities. | | Molecules. 2014 Mar 17;19(3):3173-80. | | Antibacterial effects of afzelin isolated from Cornus macrophylla on Pseudomonas aeruginosa, a leading cause of illness in immunocompromised individuals.[Pubmed: 24642906] | The crude ethyl acetate extract of the leaves of Cornus macrophylla showed antibacterial activity against Pseudomonas aeruginosa, a leading cause of illness in immunocompromised individuals. Bioactivity-guided separation led to the isolation of kaempferol 3-O-α-L-rhamnopyranoside (Afzelin).
METHODS AND RESULTS:
The structure was determined based on evaluation of its spectroscopic (UV, MS, and NMR) data. The minimum inhibitory concentration (MIC) of Afzelin against Pseudomonas aeruginosa was found to be 31 µg/mL. In addition, the results indicated that a hydroxyl group at C3 of the C-ring of the flavone skeleton and the rhamnose group may act as a negative factor and an enhancing factor, respectively, in the antibacterial activities of Afzelin. | | Arch Pharm Res. 2015 Jun;38(6):1168-77. | | Preliminary in vitro and ex vivo evaluation of afzelin, kaempferitrin and pterogynoside action over free radicals and reactive oxygen species.[Pubmed: 25315635] | Biological activities of flavonoids have been extensively reviewed in literature. The biochemical profile of Afzelin, kaempferitrin, and pterogynoside acting on reactive oxygen species was investigated in this paper.
METHODS AND RESULTS:
The flavonoids were able to act as scavengers of the superoxide anion, hypochlorous acid and taurine chloramine. Although flavonoids are naturally occurring substances in plants which antioxidant activities have been widely advertised as beneficial, Afzelin, kaempferitrin, and pterogynoside were able to promote cytotoxic effect. In red blood cells this toxicity was enhanced, depending on flavonoids concentration, in the presence of hypochlorous acid, but reduced in the presence of 2,2'-azo-bis(2-amidinopropane) free radical. These flavonoids had also promoted the death of neutrophils, which was exacerbated when the oxidative burst was initiated by phorbol miristate acetate.
CONCLUSIONS:
Therefore, despite their well-known scavenging action toward free radicals and oxidants, these compounds could be very harmful to living organisms through their action over erythrocytes and neutrophils. |
|
| In vivo: |
| Mol Med Rep. 2015 Jul;12(1):71-6. | | Afzelin attenuates asthma phenotypes by downregulation of GATA3 in a murine model of asthma.[Pubmed: 25738969] | Asthma is a serious health problem causing significant mortality and morbidity globally. Persistent airway inflammation, airway hyperresponsiveness, increased immunoglobulin E (IgE) levels and mucus hypersecretion are key characteristics of the condition. Asthma is mediated via a dominant T-helper 2 (Th2) immune response, causing enhanced expression of Th2 cytokines. These cytokines are responsible for the various pathological changes associated with allergic asthma. To investigate the anti-asthmatic potential of Afzelin, as well as the underlying mechanisms involved, its anti-asthmatic potential were investigated in a murine model of asthma.
METHODS AND RESULTS:
In the present study, BALB/c mice were systemically sensitized using ovalbumin (OVA) followed by aerosol allergen challenges. The effect of Afzelin on airway hyperresponsiveness, eosinophilic infiltration, Th2 cytokine and OVA‑specific IgE production in a mouse model of asthma were investigated. It was found that Afzelin‑treated groups suppressed eosinophil infiltration, allergic airway inflammation, airway hyperresponsiveness, OVA-specific IgE and Th2 cytokine secretion.
CONCLUSIONS:
The results of the present study suggested that the therapeutic mechanism by which Afzelin effectively treats asthma is based on reduction of Th2 cytokine via inhibition of GATA-binding protein 3 transcription factor, which is the master regulator of Th2 cytokine differentiation and production. | | Br J Pharmacol . 2017 Jan;174(2):195-209. | | Afzelin ameliorates D-galactosamine and lipopolysaccharide-induced fulminant hepatic failure by modulating mitochondrial quality control and dynamics[Pubmed: 27861739] | | Abstract
Background and purpose: Fulminant hepatic failure (FHF) is a fatal clinical syndrome that results in excessive inflammation and hepatocyte death. Mitochondrial dysfunction is considered to be a possible mechanism of FHF. Afzelin, a flavonol glycoside found in Houttuynia cordata Thunberg, has anti-inflammatory and antioxidant properties. The present study elucidated the cytoprotective mechanisms of Afzelin against D-galactosamine (GalN)/LPS induced FHF, particularly focusing on mitochondrial quality control and dynamics.
Experimental approach: Mice were administered Afzelin i.p. 1 h before receiving GalN (800 mg·kg-1 )/LPS (40 μg·kg-1 ), and they were then killed 5 h after GalN/LPS treatment.
Key results: Afzelin improved the survival rate and reduced the serum levels of alanine aminotransferase and pro-inflammatory cytokines in GalN/LPS-treated mice. Afzelin attenuated the mitochondrial damage, as indicated by diminished mitochondrial swelling and mitochondrial glutamate dehydrogenase activity in GalN/LPS-treated mice. Afzelin enhanced mitochondrial biogenesis, as indicated by increased levels of PPAR-γ coactivator 1α, nuclear respiratory factor 1 and mitochondrial transcription factor A. Afzelin also decreased the level of mitophagy-related proteins, parkin and PTEN-induced putative kinase 1. Furthermore, while GalN/LPS significantly increased the level of fission-related protein, dynamin-related protein 1, and decreased the level of fusion-related protein, mitofusin 2; these effects were attenuated by Afzelin. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)