| Kinase Assay: |
| Food Chem. 2011, 126(1):31-8. | | Antioxidant and anticholinesterase active constituents from Micromeria cilicica by radical-scavenging activity-guided fractionation[Reference: WebLink] |
METHODS AND RESULTS:
From the acetone extract of Micromeria cilicica, two new (1 and 2) and five known (3–7) compounds were obtained through radical-scavenging activity guided isolation.
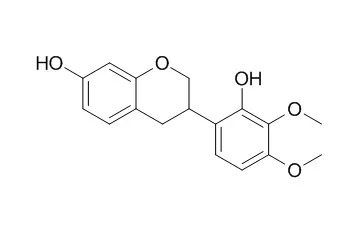
Structures of the compounds were identified as piperitone 7-O-β-d-glucoside (1), isothymonin 4′-methyl ether (2), sudachitin (3), Isomucronulatol (4), rutin (5), ursolic acid (6) and saccharose (7), based on UV, 1D-and 2D-NMR and mass spectroscopic techniques.
The antioxidant potentials of the extract and the isolated compounds were established by using three radical-scavenging assays, namely, DPPH scavenging, O2- scavenging and ABTS+ scavenging, besides β-carotene bleaching assay. Particularly, the acetone extract showed a strong inhibition of lipid peroxidation by β-carotene bleaching assay, with a result close to that of (+)-catechin.
CONCLUSIONS:
Among the pure compounds, rutin (5) showed the strongest lipid peroxidation inhibition and antiradical activity while both sudachitin (3) and Isomucronulatol (4) exhibited noticeable ABTS+ scavenging activity. The anticholinesterase activity of the compounds (1–7) was also determined. Against acetylcholinesterase, they exhibited weak inhibition while compounds 3, 4 and 6 exhibited moderate inhibition against butyrylcholinesterase. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)