| In vitro: |
| Applied Surface ence, 2007, 254(2):616-620. | | Aluminum pigment encapsulated by in situ copolymerization of styrene and maleic acid.[Reference: WebLink] |
METHODS AND RESULTS:
To improve its anticorrosion property, aluminum pigment was encapsulated by in situ copolymerization of styrene (St) and Maleic acid (MA). It was found that the conversion of monomers (C), the percentage of grafting (PG) and the grafting efficiency (GE) could attain 92%, 12%, 25%, respectively, when m(BPO)/ m(St + MA) = 10% and m(St + MA)/ m(Al) = 10%. The optimum condition for protection factor was studied according to an orthogonal testing. When m(St + MA)/ m(Al) was 20%, the encapsulated aluminum pigment simultaneously showed good anticorrosion property and luster.
CONCLUSIONS:
FTIR, SEM and particle size analysis indicated that aluminum pigment had been successfully encapsulated with styrene–Maleic acid copolymer by in situ copolymerization, which remarkably improved its anticorrosion property and the chelate complex formed between SMA and Al(III) was possibly the actual corrosion inhibitor. | | Journal of Petroleum ence & Engineering, 2010, 75(1-2):189-195. | | Maleic acid based scale inhibitors for calcium sulfate scale inhibition in high temperature application.[Reference: WebLink] | In anhydrite rich reservoir rock, calcium sulfate is one of the dominant scale components which unlike carbonate scale are not easily removable by acid or dissolver treatment.
METHODS AND RESULTS:
To inhibit calcium sulfate scale formation in high temperature producing brine water systems, Maleic acid–acrylic acid and Maleic acid–acrylamide copolymers of appropriate molecular weight were synthesized and characterized and inhibition efficiency of the copolymers was tested at static and dynamic conditions. Their iron dispersing ability measured through UV spectroscopy is found to be excellent and their biocidal activity investigated through agar culture and serial dilution technique is almost insignificant.
CONCLUSIONS:
XRD and SEM analyses proved the morphological changes of the calcium sulphate scale due to strong inhibition action. |
|
| In vivo: |
| J Clin Invest. 1999 Mar 1; 103(5): 707–713. | | Maleic acid and succinic acid in fermented alcoholic beverages are the stimulants of gastric acid secretion.[Pubmed: 10074488] | Alcoholic beverages produced by fermentation (e.g., beer and wine) are powerful stimulants of gastric acid output and gastrin release in humans. The aim of this study was to separate and specify the gastric acid stimulatory ingredients in alcoholic beverages produced by fermentation.
METHODS AND RESULTS:
Yeast-fermented glucose was used as a simple model of fermented alcoholic beverages; it was stepwise separated by different methods of liquid chromatography, and each separated solution was tested in human volunteers for its stimulatory action on gastric acid output and gastrin release. Five substances were detected by high-performance liquid chromatography and were analyzed by mass spectrometry and 1H-13C nuclear magnetic resonance spectroscopy. At the end of the separation process of the five identified substances, only the two dicarboxylic acids, Maleic acid and succinic acid, had a significant (P < 0.05) stimulatory action on gastric acid output (76% and 70% of fermented glucose, respectively), but not on gastrin release. When given together, they increased gastric acid output by 100% of fermented glucose and by 95% of maximal acid output.
CONCLUSIONS:
We therefore conclude that Maleic acid and succinic acid are the powerful stimulants of gastric acid output in fermented glucose and alcoholic beverages produced by fermentation, and that gastrin is not their mediator of action. |
|


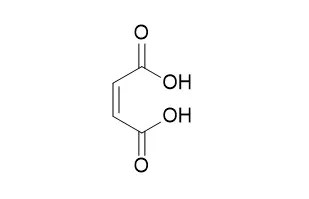



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)