| In vivo: |
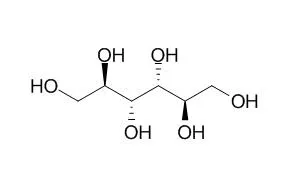
| J Neurosurg. 1985 Jul;63(1):43-8. | | Effect of mannitol on cerebral blood flow and cerebral perfusion pressure in human head injury[Pubmed: 3925092 ] | Patients with severe head injury frequently have evidence of elevated intracranial pressure (ICP) and ischemic neuronal damage at autopsy. Mannitol has been used clinically to reduce ICP with varying success, and it is possible that it is more effective in some types of head injury than in others.
METHODS AND RESULTS:
The aim of the present study was to determine the effect of Mannitol on ICP, cerebral perfusion pressure (CPP), and cerebral blood flow (CBF) in patients with severe head injury, and to discover if these effects differed in different types of injury. Measurements of CPP, ICP, and CBF were made in 55 patients with severe head injury. In general, the resting level of CBF was higher in patients with diffuse injury (mean 50.2 ml/100 gm/min) than in those with focal injury (mean 39.8 ml/100 gm/min). Mannitol consistently reduced ICP and increased CPP and CBF by 10 to 20 minutes after infusion. The lowest flows (31.8 ml/100 gm/min) were recorded from the most damaged hemispheres of patients with focal injuries and elevated ICP. The baseline levels of flow did not correlate with ICP, CPP, Glasgow Coma Scale score, or outcome. Only four of the 55 patients had a CBF of less than 20 ml/100 gm/min in either or both hemispheres.
CONCLUSIONS:
The few low CBF's in this and other studies may reflect the steady-state conditions under which measurements are made in intensive care units, and that these patients have entered a phase of reperfusion. | | Eur J Nucl Med Mol Imaging. 2017 Aug 11. | | Reduction of 68Ga-PSMA renal uptake with mannitol infusion: preliminary results.[Pubmed: 28801787] | Urea-based prostate-specific membrane antigen (PSMA) ligands labelled with 68Ga or 177Lu are new tracers with great potential for theranostic approaches in prostate cancer. However, clinical studies have shown that the kidneys are one of the off-target organs along with the salivary and lacrimal glands. In the kidneys, PSMA is physiologically expressed in the apical epithelium of the proximal tubules, and Mannitol acts as an osmotic diuretic in these tubules.
METHODS AND RESULTS:
We investigated the potential of Mannitol to reduce renal uptake of 68Ga-PSMA. Kidney uptake (SUVmax) was calculated in nine patients undergoing 68Ga-PSMA PET/CT at baseline (b-PET/CT) and after intravenous infusion of 500 ml of 10% Mannitol (m-PET/CT). Two different infusion schemes for Mannitol were used: (1) 500 ml Mannitol was infused over 40 min after 68Ga-PSMA administration (A-infusion) and (2) 250 ml Mannitol was infused over 15 min before and again after 68Ga-PSMA administration (B-infusion). In patients receiving the A-infusion, mean SUVmax increased by 11.9% and 7.4% in the right and left kidney, respectively. In patients receiving the B-infusion, mean SUVmax decreased by 24.3% and 22.4% in the right and left kidney, respectively. Our preliminary findings indicate that Mannitol may play a role in reducing off-target 68Ga-PSMA renal uptake. Administration of the osmotic diuretic should be rapid and start before 68Ga-PSMA injection.
CONCLUSIONS:
These results warrant dosimetric studies in patients treated with 177Lu-PSMA to find the best scheme for Mannitol administration. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)