| In vitro: |
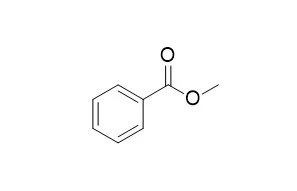
| Analytical methods, 2012, 4(12):4378-4383. | | Methyl benzoate as a non-halogenated extraction solvent for dispersive liquid–liquid microextraction: Application to the preconcentration of copper(II) with 1-nitroso-2-naphthol.[Reference: WebLink] | Methyl benzoate, whose density is greater than that of water, was used as a non-halogenated extraction solvent for dispersive liquid–liquid microextraction (DLLME).
METHODS AND RESULTS:
The quantitative extraction of some elements, such as Au(III), Cd(II), Co(II), Fe(III), Ni(II), Pb(II), and Pd(II), was achieved using some traditional extractants, including diethyldithiocarbamate, 1-(2-pyridilazo)-2-naphthol, 8-hydroxyquinoline, and 1-nitroso-2-naphthol (NN) with acetone as a disperser solvent. As a demonstration, the preconcentration of Cu(II) with NN was studied in detail. The Cu(II) in 8 mL of a sample solution was rapidly and quantitatively extracted at pH 7 using 80 μL of Methyl benzoate containing 1 mmol L−1 of NN; in this system, no disperser solvent was needed. After centrifugation, the extracted Cu(II) in a sedimented phase could be determined by electrothermal atomic absorption spectrometry. The detection limit was 2.3 ng L−1 (three times the standard deviation of the blank values, n = 8) as the Cu(II) concentration in 8 mL of the sample solution.
CONCLUSIONS:
The proposed method was applicable to the determination of Cu(II) in some water samples, including tap water, ground water, and snow-melted water without any interference. | | Journal of separation science, 2005, 28(18):2517-2525. | | Methyl benzoate as a marker for the detection of mold in indoor building materials.[Reference: WebLink] | A convenient analytical method to quantify volatile organic compounds (VOCs) emitted from various building materials has not been addressed yet. This work presents a new and rapid automated method using SPME combined with GC/MS. Methyl benzoate - as a metabolic biomarker for mold growth-was used to indicate VOCs and to determine and assess mold growth on damp samples. Gypsum board and wall-board paper were used as examples of common indoor building materials.
METHODS AND RESULTS:
Optimized extraction conditions were carried out manually, using a GC/flame ionization detector. Moldy samples were analyzed using an automated SPME-GC/MS analysis under optimized conditions. The amount of Methyl benzoate emitted from the studied samples ranged from 32 to 46 ppb, where the density of the fungal biomass was found to be 8 x 10(4) cells/mL. A relationship between the amount of fungal biomass and the emitted concentration of Methyl benzoate was found and assessed based upon cultured mold samples taken from indoor building sites. The analytical method shows promise for the compound Methyl benzoate, which can easily be identified at low detection limits (LOD = 3 ppb) and good linearity (>0.988), and its extraction and detection can be accomplished cleanly by current extraction techniques.
CONCLUSIONS:
Results suggest that this method with easy sample preparation can be used for quantitation and, of importance, minimal matrix effects are observed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)