| In vitro: |
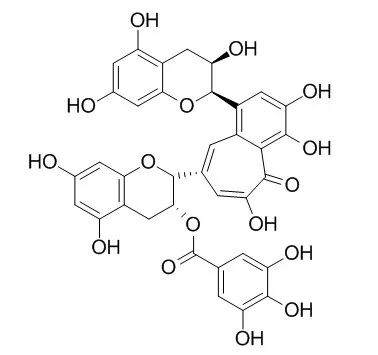
| Basic Clin Pharmacol Toxicol. 2008 Jul;103(1):66-74. | | Theaflavin-3-gallate and theaflavin-3'-gallate, polyphenols in black tea with prooxidant properties.[Pubmed: 18346048] | This study compared the in vitro responses of human gingival fibroblasts and of carcinoma cells derived from the tongue to theaflavin-3-gallate (TF-2A) and Theaflavin-3'-gallate (TF-2B), polyphenols in black tea.
METHODS AND RESULTS:
In a cell-free assay, TF-2A and Theaflavin-3'-gallate reacted directly with reduced glutathione (GSH), in a time- and concentration-dependent manner. Intracellular storages of GSH were depleted on treatment of the cells with the theaflavin monomers. Depletion of intracellular GSH was more extensive with TF-2A than with Theaflavin-3'-gallate and was more pronounced in the carcinoma, than in the normal, cells. The toxicities of the theaflavins were potentiated when the cells were cotreated with the GSH depleter, d,l-buthionine-[S,R]-sulfoximine. In the presence of catalase, pyruvate and divalent cobalt, all scavengers of reactive oxygen species, the cytotoxicities of the theaflavins were lessened. TF-2A and Theaflavin-3'-gallate induced lipid peroxidation in the carcinoma cells, whereas in the fibroblasts, peroxidation was evident upon exposure to TF-2A, but not to Theaflavin-3'-gallate.
CONCLUSIONS:
These studies demonstrated that the black tea theaflavin monomers, TF-2A and Theaflavin-3'-gallate, act as prooxidants and induce oxidative stress, with carcinoma cells more sensitive than normal fibroblasts. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)