| Animal Research: |
| Pharmacological Research, 2005, 52(6):467-474. | | Antioxidant property of alpha-asarone against noise-stress-induced changes in different regions of rat brain.[Reference: WebLink] | Free radicals and other reactive species are considered to be an important causative factor in the development of neurodegenerative diseases. Recent reports have indicated that exposure to loud noise generates excess oxygen free radicals (OFR) in the brain.
Antioxidant properties of medicinal plants are attracting more and more research in medicine, to counteract OFR and to minimize the neurodegenerative processes.
METHODS AND RESULTS:
The drug alpha-Asarone (3, 6 and 9 mg kg(-1) body weight, i.p., for 30 days), one of the active principle components of Acorus calamus Linn., was administered intraperitoneally 1/2 h before the animals were exposed to noise-stress (100 dB for 4 h d(-1), for 30 days). We investigated whether 30 days exposure of noise can produce an oxidative stress. Further, if yes then, could alpha-Asarone counteract the stress. This was verified by measuring the activity of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), levels of reduced glutathione (GSH), Vitamin C, Vitamin E, protein thiols and lipid peroxidation (LPO) in different regions of the rat brain. All the three doses of alpha-Asarone had an effectively protective role by normalizing the increased SOD and LPO, decreased CAT, GPx, GSH, Vitamins C and E and protein thiols due to noise exposure. Thus, action of alpha-Asarone against noise-stress may be due its antioxidant property.
CONCLUSIONS:
Our data proved that antioxidant property of alpha-Asarone against noise-stress induced changes in the rat brain. Further, more clinical studies are required to investigate effectiveness of the alpha-Asarone in noisy environment in human subjects. | | European Journal of Pharmacology, 2017:S001429991730674X. | | Alpha-asarone attenuates depression-like behavior in nicotine-withdrawn mice: Evidence for the modulation of hippocampal pCREB levels during nicotine-withdrawal.[Pubmed: 29042206] | In the present study, the effect alpha-Asarone on nicotine withdrawal-induced depression-like behavior in mice was investigated.
METHODS AND RESULTS:
In this study, mice were exposed to drinking water or nicotine solution (10–200 µg/ml) as a source of drinking for forty days. During this period, daily fluid consumption, food intake and body weight were recorded. The serum cotinine level was estimated before nicotine withdrawal. Naïve mice or nicotine-withdrawn mice were treated with alpha-Asarone
(5, 10 and 20 mg/kg, i.p.) or bupropion (10 mg/kg, i.p.) for eight consecutive days and the forced swim test (FST) or locomotor activity test was conducted. In addition, the effect of alpha-Asarone
or bupropion on the hippocampal pCREB, CREB and BDNF levels during nicotine-withdrawal were measured. Results indicated that alpha-Asarone (5, 10 and 20 mg/kg, i.p.) or bupropion (10 mg/kg, i.p.) pretreatment did not significantly alter the immobility time in the FST or spontaneous locomotor activity in naïve mice. However, the immobility time of nicotine-withdrawn mice was significantly attenuated with alpha-Asarone (5, 10 and 20 mg/kg, i.p.) or bupropion (10 mg/kg, i.p.) pretreatment in the FST. Besides, alpha-Asarone
(5, 10 and 20 mg/kg, i.p.) or bupropion (10 mg/kg, i.p.) pretreatment significantly attenuated the hippocampal pCREB levels in nicotine-withdrawn mice.
CONCLUSIONS:
Overall, the present results indicate that alpha-Asarone
treatment attenuated the depression-like behavior through the modulation of hippocampal pCREB levels during nicotine-withdrawal in mice. |
|


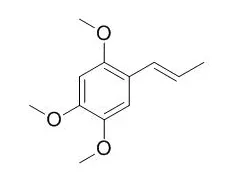



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)