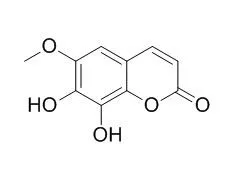
| Description: |
Fraxetin has dual-antioxidative ,hepatoprotective and antihyperglycemic functions, it shows potent protective effects against CCl4 induced oxidative stress and hepatic fibrosis, has a marked inhibitory effect on S.aureus proliferation. It increased the level of Nrf2/ARE, and HO-1, inhibit the formation of ROS, cytochrome c release, activation of caspase-3 and 9, and suppressed the up-regulation of Bax.
|
| Targets: |
Nrf2 | LDL | ROS | Caspase | HO-1 | Bcl-2/Bax |
| In vitro: |
| Mol Med Rep. 2014 Nov;10(5):2341-5. | | Antibacterial mechanism of fraxetin against Staphylococcus aureus.[Pubmed: 25189268] | Fraxetin is one of the main constituents of the traditional medicinal plant Fraxinus rhynchophylla. The inhibitory effect of Fraxetin on various bacterial strains has been extensively reported, however, its mechanism of action on bacterial cells remains to be elucidated.
METHODS AND RESULTS:
In the present study, the antibacterial mechanism of Fraxetin on Staphylococcus aureus was systematically investigated by examining its effect on cell membranes, protein synthesis, nucleic acid content and topoisomerase activity. The results indicated that Fraxetin increased the permeability of the cell membrane but did not render it permeable to macromolecules, such as DNA and RNA. Additionally, the quantity of protein, DNA and RNA decreased to 55.74, 33.86 and 48.96%, respectively following treatment with Fraxetin for 16 h. The activity of topoisomerase I and topoisomerase II were also markedly inhibited as Fraxetin concentration increased. The result of the ultraviolet‑visible spectrophotometry demonstrated that the DNA characteristics exhibited a blue shift and hypochromic effect following treatment with Fraxetin.
CONCLUSIONS:
These results indicated that Fraxetin had a marked inhibitory effect on S.aureus proliferation. Further mechanistic studies showed that Fraxetin could disrupt nucleic acid and protein synthesis by preventing topoisomerase from binding to DNA. | | Neurosci Res. 2005 Sep;53(1):48-56. | | Fraxetin prevents rotenone-induced apoptosis by induction of endogenous glutathione in human neuroblastoma cells.[Pubmed: 15996779 ] | Fraxetin belongs to an extensive group of natural phenolic anti-oxidants.
METHODS AND RESULTS:
In the present study, using a human neuroblastoma SH-SY5Y cells, we have investigated the protective effects of this compound on modifications in endogenous reduced glutathione (GSH), intracellular oxygen species (ROS) and apoptotic death on rotenone-mediated cytoxicity. Incubation of cells with the Fraxetin led to a significant elevation dose-dependent of cellular GSH and this was accompanied by a marked protection against rotenone-mediated toxicity, which was also significantly reversed in the cells with buthionine sulfoximine (BSO) co-treatment. Taken together, this study suggested that intracellular GSH appeared to be an important factor in Fraxetin-mediated cytoprotection against rotenone-toxicity in SH-SY5Y cells. Fraxetin at 10-100 muM inhibited the formation of ROS, cytochrome c release, activation of caspase-3 and 9, and suppressed the up-regulation of Bax, whereas no significant change occurred in Bcl-2 levels.
CONCLUSIONS:
Our results indicated that the anti-oxidative and anti-apoptotic properties render this natural compound potentially protective against rotenone-induced cytotoxicity. |
|
| In vivo: |
| Phytomedicine. 2014 Feb 15;21(3):240-6. | | Antioxidant and intestinal anti-inflammatory effects of plant-derived coumarin derivatives.[Pubmed: 24176844] | Coumarins, also known as benzopyrones, are plant-derived products with several pharmacological properties, including antioxidant and anti-inflammatory activities. Based on the wide distribution of coumarin derivatives in plant-based foods and beverages in the human diet, our objective was to evaluate both the antioxidant and intestinal anti-inflammatory activities of six coumarin derivatives of plant origin (scopoletin, scoparone, Fraxetin, 4-methyl-umbeliferone, esculin and daphnetin) to verify if potential intestinal anti-inflammatory activity was related to antioxidant properties.
METHODS AND RESULTS:
Intestinal inflammation was induced by intracolonic instillation of TNBS in rats. The animals were treated with coumarins by oral route. The animals were killed 48 h after colitis induction. The colonic segments were obtained after laparotomy and macroscopic and biochemical parameters (determination of glutathione level and myeloperoxidase and alkaline phosphatase activities) were evaluated. The antioxidant properties of these coumarins were examined by lipid peroxidation and DPPH assays.
Treatment with esculin, scoparone and daphnetin produced the best protective effects. All coumarin derivatives showed antioxidant activity in the DPPH assay, while daphnetin and Fraxetin also showed antioxidant activity by inhibiting lipid peroxidation. Coumarins, except 4-methyl-umbeliferone, also showed antioxidant activity through the counteraction of glutathione levels or through the inhibition of myeloperoxidase activity.
CONCLUSIONS:
The intestinal anti-inflammatory activity of coumarin derivatives were related to their antioxidant properties, suggesting that consumption of coumarins and/or foods rich in coumarin derivatives, particularly daphnetin, esculin and scoparone, could prevent intestinal inflammatory disease. | | Biochimie. 2013 Oct;95(10):1848-54. | | Antihyperglycemic effect of fraxetin on hepatic key enzymes of carbohydrate metabolism in streptozotocin-induced diabetic rats.[Pubmed: 23806420] | Epidemiological studies have demonstrated that the diabetes mellitus is a serious health burden for both governments and healthcare providers.
METHODS AND RESULTS:
The present study was hypothesized to evaluate the antihyperglycemic potential of Fraxetin by determining the activities of key enzymes of carbohydrate metabolism in streptozotocin (STZ) - induced diabetic rats. Diabetes was induced in male albino Wistar rats by intraperitoneal administration of STZ (40 mg/kg b.w). Fraxetin was administered to diabetic rats intra gastrically at 20, 40, 80 mg/kg b.w for 30 days. The dose 80 mg/kg b.w, significantly reduced the levels of blood glucose and glycosylated hemoglobin (HbA1c) and increased plasma insulin level. The altered activities of the key enzymes of carbohydrate metabolism such as glucokinase, glucose-6-phosphate dehydrogenase, glucose-6-phosphatase, fructose-1,6-bisphosphatase and hepatic enzymes (aspartate transaminase (AST), alanine transaminase (ALT) and alkaline phosphatase (ALP)) in the liver tissues of diabetic rats were significantly reverted to near normal levels by the administration of Fraxetin. Further, Fraxetin administration to diabetic rats improved body weight and hepatic glycogen content demonstrated its antihyperglycemic potential.
CONCLUSIONS:
The present findings suggest that Fraxetin may be useful in the treatment of diabetes even though clinical studies to evaluate this possibility may be warranted. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)