Natural Products

- ChemFaces is a professional high-purity natural products manufacturer.
- Product Intended Use
- 1. Reference standards
- 2. Pharmacological research
- 3. Inhibitors



| Size /Price /Stock | 10 mM * 1 mL in DMSO / $52.6 / In-stock | Other Packaging | *Packaging according to customer requirements(100uL/well, 200uL/well and more), and Container use Storage Tube With Screw Cap |

| Size /Price /Stock | 10 mM * 100 uL in DMSO / Inquiry / In-stock 10 mM * 1 mL in DMSO / Inquiry / In-stock | Related Libraries |
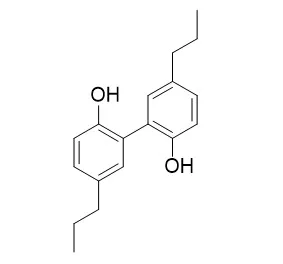
| Description: | Tetrahydromagnolol can activate cannabinoid (CB) receptors. |
| Targets: | Cannabinoid Receptor | GPR | GABA Receptor |
| Source: | The barks of Magnolia officinalis |
| Solvent: | Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| Storage: | Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to 24 months(2-8C). Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour. Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com |
| After receiving: | The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling. |
 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6982 mL | 18.4911 mL | 36.9822 mL | 73.9645 mL | 92.4556 mL |
| 5 mM | 0.7396 mL | 3.6982 mL | 7.3964 mL | 14.7929 mL | 18.4911 mL |
| 10 mM | 0.3698 mL | 1.8491 mL | 3.6982 mL | 7.3964 mL | 9.2456 mL |
| 50 mM | 0.074 mL | 0.3698 mL | 0.7396 mL | 1.4793 mL | 1.8491 mL |
| 100 mM | 0.037 mL | 0.1849 mL | 0.3698 mL | 0.7396 mL | 0.9246 mL |
| Citation [1] PLoS One. 2013 Oct 30;8(10):e77739. | The natural product magnolol as a lead structure for the development of potent cannabinoid receptor agonists.[Pubmed: 24204944 ] |
| Magnolol (4-allyl-2-(5-allyl-2-hydroxyphenyl)phenol), the main bioactive constituent of the medicinal plant Magnolia officinalis, and its main metabolite Tetrahydromagnolol were recently found to activate cannabinoid (CB) receptors. We now investigated the structure-activity relationships of (tetrahydro)magnolol analogs with variations of the alkyl chains and the phenolic groups and could considerably improve potency. Among the most potent compounds were the dual CB1/CB2 full agonist 2-(2-methoxy-5-propyl-phenyl)-4-hexylphenol (61a, K(i) CB1:0.00957 μM; K(i) CB2:0.0238 μM), and the CB2-selective partial agonist 2-(2-hydroxy-5-propylphenyl)-4-pentylphenol (60, K(i) CB1:0.362 μM; K(i ) CB2:0.0371 μM), which showed high selectivity versus GPR18 and GPR55. Compound 61b, an isomer of 61a, was the most potent GPR55 antagonist with an IC50 value of 3.25 μM but was non-selective. The relatively simple structures, which possess no stereocenters, are easily accessible in a four- to five-step synthetic procedure from common starting materials. The central reaction step is the well-elaborated Suzuki-Miyaura cross-coupling reaction, which is suitable for a combinatorial chemistry approach. The scaffold is versatile and may be fine-tuned to obtain a broad range of receptor affinities, selectivities and efficacies. | |
| Citation [2] ACS Med Chem Lett. 2012 Nov 14;4(1):41-5. | Magnolia Extract, Magnolol, and Metabolites: Activation of Cannabinoid CB2 Receptors and Blockade of the Related GPR55.[Pubmed: 24900561 ] |
| The bark of Magnolia officinalis is used in Asian traditional medicine for the treatment of anxiety, sleeping disorders, and allergic diseases. We found that the extract and its main bioactive constituents, magnolol and honokiol, can activate cannabinoid (CB) receptors. In cAMP accumulation studies, magnolol behaved as a partial agonist (EC50 = 3.28 μM) with selectivity for the CB2 subtype, while honokiol was less potent showing full agonistic activity at CB1 and antagonistic properties at CB2. We subsequently synthesized the major metabolites of magnolol and found that Tetrahydromagnolol (7) was 19-fold more potent than magnolol (EC50 CB2 = 0.170 μM) exhibiting high selectivity versus CB1. Additionally, 7 behaved as an antagonist at GPR55, a CB-related orphan receptor (K B = 13.3 μM, β-arrestin translocation assay). Magnolol and its metabolites may contribute to the biological activities of Magnolia extract via the observed mechanisms of action. Furthermore, the biphenylic compound magnolol provides a simple novel lead structure for the development of agonists for CB receptors and antagonists for the related GPR55. | |
| Citation [3] Hum Psychopharmacol. 2017 May;32(3). | Is there a potential of misuse for Magnolia officinalis compounds/metabolites?[Pubmed: 28517911 ] |
| Magnolia bark contains magnolol, metabolized to Tetrahydromagnolol and honokiol, with both GABA-ergic/cannabimimetic activities, hence of possible attraction to vulnerable individuals/recreational misusers. METHODS: A literature review, assessment of related anecdotal online Magnolia misuse's reports and an overview of Magnolia products' online acquisition possibilities has been here described. RESULTS: No peer-reviewed papers about Magnolia abuse/misuse/dependence/addiction were identified. Conversely, from a range of websites emerged potentially 3 groups of Magnolia misusers: (a) subjects with a psychiatric history already treated with benzodiazepines, being attracted to Magnolia bark as a "natural sedative"; (b) polydrug misusers, ingesting Magnolia with a range of other herbs/plants, attracted by the GABA-ergic/cannabimimetic activities; (c) subjects naive to the misusing drugs' scenario, perceiving Magnolia as a natural dietary supplement/weight-control compound. CONCLUSIONS: To the best of our knowledge, this is the first paper commenting on the possible Magnolia derivatives' potential of misuse. Magnolia's recent increase in popularity, mainly as a sedative, may be arguably due to its peculiar pharmacological properties/acceptable affordability levels/virtually worldwide favorable legal status and customers' attraction to a product being perceived as "natural" and hence somehow "safe." Future/potent/synthetic magnolol and honokiol structural analogues could however contribute to increasing the number of synthetic GABA-ergic/cannabimimetic misusing compounds. |