| Free Radic Biol Med . 2019 Apr;134:598-603 |
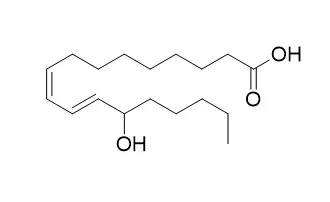
| 13-HODE, 9-HODE and ALOX15 as potential players in Rett syndrome OxInflammation[Pubmed: 30743046] |
| Mutations in the MECP2 gene are the main cause of Rett syndrome (RTT), a pervasive neurodevelopmental disorder, that shows also multisystem disturbances associated with a metabolic component. The aim of this study was to investigate whether an increased production of oxidized linoleic acid metabolites, specifically 9- and 13-hydroxyoctadecadienoic acids (HODEs), can contribute to the altered the redox and immune homeostasis, suggested to be involved in RTT. Serum levels of 9- and 13-HODEs were elevated in RTT and associated with the expression of arachidonate 15-Lipoxygenase (ALOX15) in peripheral blood mononuclear cells (PBMCs). Omega-3 polyunsaturated fatty acids supplementation has shown to lower HODEs levels in RTT. Statistically significant correlation was demonstrated between the increased plasma HODEs levels and the lipoprotein-associated phospholipase A2 (Lp-PLA2) activity. Collectively, these findings reinforce the concept of the key role played by lipid peroxidation in RTT, and the possible ability of omega-3 polyunsaturated fatty acids supplementation in improving the oxinflammation status in RTT. |
| Wien Klin Wochenschr . 1991;103(14):416-21. |
| 13-Hydroxyoctadecadienoic acid (13-HODE) metabolism and endothelial cell adhesion molecule expression: effect on platelet vessel wall adhesion[Pubmed: 1718092] |
| Endothelial cells synthesize two important fatty acid metabolites, PGI2, which is synthesized from arachidonic acid via the cyclooxygenase pathway, and 13-HODE, which is synthesized from linoleic acid via the lipoxygenase pathway. PGI2 is synthesized following cell activation or injury while 13-HODE is synthesized in the unstimulated cell. While the role of PGI2 in platelet vessel wall interactions has been studied extensively, the role of 13-HODE in platelet vessel wall interactions is just now being understood. The present evidence suggests that 13-HODE is continuously synthesized in "resting" vessel wall cells and is in close juxtaposition with the ubiquous integrin adhesion molecule, the vitronectin receptor. The observation that the endothelial cell is not adhesive when 13-HODE and the vitronectin receptor are in close association and becomes adhesive when these two moieties dissociate and the vitronectin receptor relocates on the surface of the cell, provides further evidence that 13-HODE may induce conformational changes in the vitronectin receptor to reduce its ability to recognize its adhesive ligands. The additional observations that 13-HODE levels in both human and animal vessel walls are inversely related with vessel wall adhesivity, and that this adhesivity can be altered by altering 13-HODE synthesis, provides evidence that 13-HODE down-regulates the thrombogenecity of the injured vessel wall surface. |
| Adv Exp Med Biol . 1997;433:291-4. |
| The importance of linoleic acid metabolites in cancer metastasis and in the synthesis and actions of 13-HODE[Pubmed: 9561154] |
| Large scale human epidemiological studies indicate that high intakes of linoleic acid protect against the development of cancer. One mechanism may be the generation of 13-HODE from linoleic acid. 13-HODE prevents cell adhesion to endothelial cells and can inhibit cancer metastasis. 13-HODE synthesis is enhanced by cyclic AMP. Gamma-linolenic acid, a desaturated metabolite of linoleic acid, causes substantial stimulation of 13-HODE synthesis. A fall in gamma-linolenic acid synthesis with age may be related to the age-related fall in 13-HODE formation. |
| Cancer Epidemiol Biomarkers Prev . 1996 Jan;5(1):53-6. |
| Decreased levels of 13-hydroxyoctadecadienoic acid (13-HODE) dehydrogenase in neoplastic tissue of human colon biopsies[Pubmed: 8770467] |
| Recent studies have identified a role for the oxidation product of linoleic acid, 13-hydroxyoctadecadienoic acid (13-HODE) in cell proliferation. The enzyme 13-HODE dehydrogenase catalyzes the conversion of 13-HODE to 13-oxooctadecadienoic acid. This enzyme has been shown to correlate with the degree of differentiation of intestinal cells in both in vitro and in vivo models. Higher enzyme levels are found in more differentiated cell types. The present study was done to determine if enzyme levels of 13-HODE dehydrogenase are predictive of the differentiation status of biopsies from human colonic mucosa. Twenty-eight patients who underwent diagnostic colonoscopy (10 patients with adenocarcinoma and 18 with adenomatous polyps) had biopsies taken from both normal rectal mucosa and neoplastic mucosa. The determination of 13-HODE dehydrogenase activity was conducted by high-performance liquid chromatography analysis of all biopsy samples. Sixteen of the 18 patients with polyps had lower 13-HODE dehydrogenase activity in the adenoma than in the uninvolved rectal mucosa (P = 0.001). The colon adenocarcinomas also had less 13-HODE dehydrogenase activity in the cancer biopsy tissue than in uninvolved rectal mucosa (P = 0.041) These data are consistent with a role for 13-HODE dehydrogenase in intestinal cell differentiation. Understanding the precise role of this enzymatic reaction could be important potentially in the therapy and biology of colon cancer. In addition, measurements of 13-HODE dehydrogenase may be a useful parameter by which to ascertain the differentiation status of intestinal cells in vitro. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)