| In vivo: |
| Journal of Immunology, 2014, 193(6):2772. | | 6-Methoxyflavone inhibits NFAT translocation into the nucleus and suppresses T cell activation.[Pubmed: 25114106] | NFAT plays a crucial role in the immune system by regulating the transcription of inducible genes during immune responses. In T cells, NFAT proteins govern various cellular events related to T cell development, activation, tolerance induction, and differentiation. We previously reported the NFAT1-dependent enhancer activity of conserved noncoding sequence (CNS)-9, a distal cis-acting element, in the regulation of IL-10 transcription in T cells.
METHODS AND RESULTS:
In this study, we developed a T cell-based reporter system to identify compounds that modulate the regulatory activity of CNS-9. Among the identified candidates, 6-Methoxyflavone (6-MF) significantly inhibited the enhancer activity of CNS-9, thereby reducing IL-10 expression in T cells without affecting cell viability. 6-MF also downregulated the transcription of NFAT1 target genes such as IL-4, IL-13, and IFN-γ. Treatment of 6-MF inhibited the translocation of NFAT1 into the nucleus, which consequently interrupted NFAT1 binding to the target loci, without affecting the expression or dephosphorylation of NFAT1. Treatment of 6-MF to CD4(+) T cells or B cells isolated from mice with atopic dermatitis significantly reduced disease-associated cytokine production, as well as the levels of IgE. In addition, oral administration of 6-MF to atopic dermatitis mice ameliorated disease symptoms by reducing serum IgE levels and infiltrating lymphocytes.
CONCLUSIONS:
Conclusively, our results suggest that 6-MF can be a potential candidate for the development of an effective immunomodulator via the suppression of NFAT-mediated T cell activation. |
|


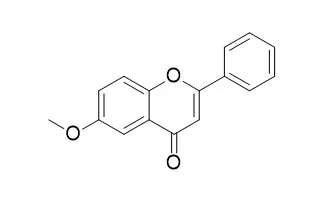



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)