| In vitro: |
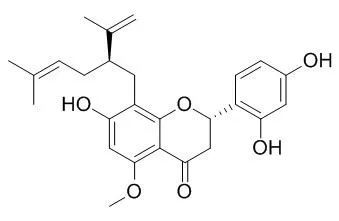
| J. Nat.Prod., 2004, 67(11):1829-32. | | Estrogenic and anticarcinogenic properties of kurarinone, a lavandulyl flavanone from the roots of Sophora flavescens.[Pubmed: 15568770 ] | Kurarinone, a lavandulyl flavanone, was isolated from a polyphenolic extract of the roots of Sophora flavescens using fractionation guided by estrogenic activity, which was determined by recombinant yeast and Ishikawa Var-I bioassays.
METHODS AND RESULTS:
Kurarinone showed weak estrogenic activity both in the yeast screen and in the Ishikawa Var-I assay with EC(50) values of 4.6 and 1.66 microM, respectively. Furthermore, Kurarinone was found to have potent cytotoxic activity (IC(50) value = 22.2 microM) against human MCF-7/6 breast cancer cells in the sulforhodamine-B assay. |
|
| In vivo: |
| Biochem. Pharmacol., 2013, 85(8):1134-44. | | Kurarinone regulates immune responses through regulation of the JAK/STAT and TCR-mediated signaling pathways.[Pubmed: 23333426 ] | Sophora flavescens is a medicinal herb that contains flavonoids and quinolizidine alkaloids and has a wide range of biological activities due to its anti-inflammatory, anti-bacterial and anti-cancer properties. We isolated a series of flavonoids from the roots of Sophora flavescens and examined their ability to inhibit immune responses.
METHODS AND RESULTS:
Among the flavonoids, Kurarinone exhibited the strongest inhibitory effect on immune responses. Kurarinone suppressed the differentiation of CD4(+) T cells by inhibiting the expression and production of T-cell lineage-specific master regulators and cytokines. Our results also demonstrated that Kurarinone directly suppressed the cytokine-induced Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling and T-cell receptor (TCR) pathways. In two established animal models of chronic inflammatory skin disease, one in which psoriasis-like skin disease was induced by an interleukin 23 (IL-23) injection into mouse ears and another in which 2,4,6-trinitrochlorobenzene (TNCB) application on the abdomens of mice was used to induce contact dermatitis, Kurarinone repressed disease development by inhibiting the expression of pro-inflammatory mediators, including cytokines, chemokines and enzyme in murine ear skin.
CONCLUSIONS:
This study provides new evidence that Kurarinone may ameliorate chronic inflammatory skin diseases through the suppression of pathogenic CD4(+) T-cell differentiation and the overall immune response. | | Int Immunopharmacol . 2018 Sep;62:227-236. | | The flavonoid kurarinone inhibits clinical progression of EAE through inhibiting Th1 and Th17 cell differentiation and proliferation[Pubmed: 30031314] | | Abstract
Introduction: The flavonoid Kurarinone suppresses CD4+ T-cell-mediated chronic inflammatory dermatitis. However, Kurarinone's effects upon autoimmune central nervous system (CNS) disease remain unknown. We investigated the potential therapeutic effects and molecular mechanism(s) of Kurarinone in an experimental autoimmune encephalomyelitis (EAE) murine model of multiple sclerosis (MS).
Materials and methods: Myelin oligodendrocyte glycoprotein (MOG35-55) peptide-induced EAE was constructed in wild-type mice. Effects of Kurarinone (100 mg/kg/day) upon clinical scores were assessed based on physical traits and signs. Spinal cord sections were extracted to assess inflammation, demyelination, and mRNA expression of key pro-inflammatory cytokines and chemokines. CNS-infiltrating mononuclear cells (MNCs) and splenocytes were harvested; flow cytometry was then applied to determine CD4+ and CD8+ T-cell percentages as well as Th1/Th2/Th17 subset percentages. Purified naïve CD4+ T-cells underwent in vitro T-cell polarization and proliferation to assess Kurarinone's effects.
Results: Prophylactic and treatment regimens of Kurarinone significantly improved clinical scores in the MOG35-55 peptide-induced EAE model (P < 0.05). Kurarinone significantly lowered CNS inflammation and demyelination (61% and 83% decreases, respectively; P < 0.05), significantly decreased MNC infiltration into CNS tissue (42% decrease; P < 0.05), and significantly inhibited levels of several pro-inflammatory cytokines and chemokines (P < 0.05). Kurarinone significantly lowered CD4+ and CD8+ CNS T-cell counts (51% and 80% decreases, respectively; P < 0.05) and significantly reduced CNS Th1 and Th17 cell percentages (24% and 44% decreases, respectively; P < 0.05). Kurarinone significantly inhibited in vitro Th1, Th2, and Th17 cell differentiation and proliferation (P < 0.05).
Conclusions: Kurarinone significantly inhibits the clinical progression of EAE through the inhibition of Th1 and Th17 cell differentiation and proliferation. Kurarinone may show promise as an immunomodulatory therapeutic agent in treating MS.
Keywords: Encephalomyelitis; Flavonoid; Kurarinone; Multiple sclerosis. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)