| In vitro: |
| Anaerobe. 2004 Oct;10(5):269-76. | | Myristic acid supports the immediate inhibitory effect of lauric acid on ruminal methanogens and methane release.[Pubmed: 16701527] |
METHODS AND RESULTS:
Two in vitro experiments were carried out with the Hohenheim gas test (HGT) apparatus in order to investigate dose-dependent effects and interactions of non-esterified lauric acid (C(12)) and Myristic acid (C(14)) given either individually or in mixture on ruminal methanogens and methanogenesis. Special emphasis was also put on the relationship between effects on methane formation and methanogenic counts. The in vitro incubations were conducted in 10mL ruminal fluid and 20mL buffer solution and lasted for 24h. In the first experiment, 14 levels of C(12), C(14) and stearic acid (C(18); control) were supplied each in increasing steps of 2.5mg covering the range from 0 to 32.5mg. In the second experiment, dosages ranging from 2.5 to 30mg C(12) were supplemented in steps of 2.5mg either without or with 10, 20 or 30mg of C(14). Counts of total Archaea and individual methanogenic orders were determined by the fluorescence in situ hybridization technique using 16S rRNA oligonucleotide probes. In experiment 1, a methane-suppressing effect of more than 80% was achieved with a supply of 30mg C(12), whereas C(14) and C(18) had no effect. Incubation liquid counts of total Archaea and individual methanogenic orders (Methanococcales, Methanosarcinales, Methanomicrobiales and Methanobacteriales) exponentially decreased as a response to C(12) and C(14) to about the same degree (up to 90%) and, to a lesser extent, by C(18). The proportions of the orders of total methanogenic population were not altered by any of the fatty acids. In experiment 2, an additional supply of 10 or 20mg of C(14) supported the suppression of methanogenesis and methanogens by C(12) synergistically. Supplementing 30mg instead of 20mg of C(14) did not further increase the efficacy of C(12) in suppressing methane formation and methanogens.
CONCLUSIONS:
The study illustrated the advantage of using mixtures of C(12) and C(14) in ruminant nutrition to suppress methane emission since mixtures will reduce the amounts of the less palatable C(12) required in feed. | | Biochimie, 2007, 89(12):1553-1561. | | Myristic acid increases the activity of dihydroceramide Δ4-desaturase 1 through its N-terminal myristoylation[Pubmed: 17716801] | Dihydroceramide Δ4-desaturase (DES) catalyzes the desaturation of dihydroceramide into ceramide. In mammals, two gene isoforms named DES1 and DES2 have recently been identified. The regulation of these enzymes is still poorly understood.
This study was designed to examine the possible N-myristoylation of DES1 and DES2 and the effect of this co-translational modification on dihydroceramide Δ4-desaturase activity.
METHODS AND RESULTS:
N-MyristoylTransferases (NMT) catalyze indeed the formation of a covalent linkage between myristoyl-CoA and the N-terminal glycine of candidate proteins, as found in the sequence of DES proteins. The expression of both rat DES in COS-7 cells evidenced first that DES1 but not DES2 was associated with an increased dihydroceramide Δ4-desaturase activity. Then, we showed that recombinant DES1 was myristoylated in vivo when expressed in COS-7 cells. In addition, in vitro myristoylation assay with a peptide substrate corresponding to the N-terminal sequence of the protein confirmed that NMT1 has a high affinity for DES1 myristoylation motif (apparent Km = 3.92 μM). Compared to an unmyristoylable mutant form of DES1 (Gly replaced by an Ala), the dihydroceramide Δ4-desaturase activity of the myristoylable DES1-Gly was reproducibly and significantly higher. Finally, the activity of wild-type DES1 was also linearly increased in the presence of increased concentrations of Myristic acid incubated with the cells. These results demonstrate that DES1 is a newly discovered myristoylated protein. This N-terminal modification has a great impact on dihydroceramide Δ4-desaturase activity.
CONCLUSIONS:
These results suggest therefore that Myristic acid may play an important role in the biosynthesis of ceramide and in sphingolipid metabolism. |
|
| In vivo: |
| Br J Nutr. 2002 Mar;87(3):199-210. | | Dietary myristic acid modifies the HDL-cholesterol concentration and liver scavenger receptor BI expression in the hamster.[Pubmed: 12064328 ] | The influence of Myristic acid in a narrow physiological range (0.5 to 2.4% of total dietary energy) on the plasma and hepatic cholesterol metabolism was investigated in the hamster.
METHODS AND RESULTS:
The hamsters were fed on a diet containing 12.5 g fat/100 g and 0.05 g cholesterol/100 g with 0.5% Myristic acid (LA diet) for 3 weeks (pre-period). During the following 3 weeks (test period), they were divided into four dietary groups with 0.5% (LA), 1.2% (LM), 1.8% (ML) or 2.4% (M) Myristic acid. Finally, half the hamsters in each group were again fed the LA diet for another 3 weeks (post-period). At the end of the test period, the hepatic expression of the scavenger receptor BI (SR-BI) was lower in the LM, ML and M groups than in the LA group whereas the hepatic cholesteryl ester concentration was higher. Cholesterol 7alpha hydroxylase activity was lower in the ML and M groups than in the LA and LM groups while the sterol 27 hydroxylase and 3-hydroxy-3-methyl glutaryl coenzyme A reductase activities were not modulated by dietary Myristic acid. This is the first time a negative correlation has been observed between the HDL-cholesterol concentration and the hepatic mass of SR-BI (r -0.69; P<0.0001) under physiological conditions. An inverse linear regression was also shown between SR-BI and the percentage of Myristic acid in the diet (r -0.75; P<0.0001). The hepatic mass of SR-BI in the M group had increased at the end of the post-period compared with the test-period values.
CONCLUSIONS:
The present investigation shows that Myristic acid modulates HDL-cholesterol via a regulation of the SR-BI expression. |
|


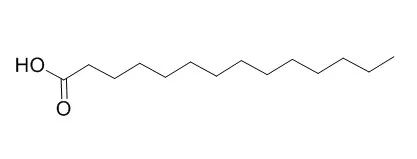



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)