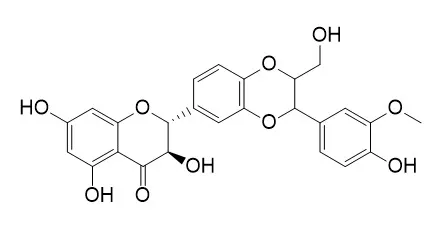
| Description: |
Silymarin possesses hepatoprotective, antioxidant, anti-inflammatory and immunomodulatory properties. It is an effective anti-cancer and chemopreventive agent, has been shown to exert multiple effects on cancer cells, including inhibition of both cell proliferation and migration. Silymarin induces apoptosis primarily through a p53-dependent pathway involving Bcl-2/Bax, cytochrome c release, and caspase activation. It inhibits PGE2 -induced cell migration through inhibition of EP2 signaling pathways (G protein dependent PKA-CREB and G protein-independent Src-STAT3).
 |
| Targets: |
PGE | PKA | Src | STAT | ROS | Bcl-2/Bax | Caspase | p53 |
| In vitro: |
| Mol Carcinog. 2015 Mar;54(3):216-28. | | Silymarin suppresses the PGE2 -induced cell migration through inhibition of EP2 activation; G protein-dependent PKA-CREB and G protein-independent Src-STAT3 signal pathways.[Pubmed: 24127286] | Silymarin has been known as a chemopreventive agent, and possesses multiple anti-cancer activities including induction of apoptosis, inhibition of proliferation and growth, and blockade of migration and invasion. However, whether Silymarin could inhibit prostaglandin (PG) E2 -induced renal cell carcinoma (RCC) migration and what are the underlying mechanisms are not well elucidated.
METHODS AND RESULTS:
Here, we found that Silymarin markedly inhibited PGE2 -stimulated migration. PGE2 induced G protein-dependent CREB phosphorylation via protein kinase A (PKA) signaling, and PKA inhibitor (H89) inhibited PGE2 -mediated migration. Silymarin reduced PGE2 -induced CREB phosphorylation and CRE-promoter activity. PGE2 also activated G protien-independent signaling pathways (Src and STAT3) and Silymarin reduced PGE2 -induced phosphorylation of Src and STAT3. Inhibitor of Src (Saracatinib) markedly reduced PGE2 -mediated migration. We found that EP2, a PGE2 receptor, is involved in PGE2 -mediated cell migration. Down regulation of EP2 by EP2 siRNA and EP2 antagonist (AH6809) reduced PGE2 -inudced migration. In contrast, EP2 agonist (Butaprost) increased cell migration and Silymarin effectively reduced butaprost-mediated cell migration. Moreover, PGE2 increased EP2 expression through activation of positive feedback mechanism, and PGE2 -induced EP2 expression, as well as basal EP2 levels, were reduced in Silymarin-treated cells.
CONCLUSIONS:
Taken together, our study demonstrates that Silymarin inhibited PGE2 -induced cell migration through inhibition of EP2 signaling pathways (G protein dependent PKA-CREB and G protein-independent Src-STAT3). |
|
| In vivo: |
| Pharmacol Rep. 2014 Oct;66(5):788-98. | | Silymarin liposomes improves oral bioavailability of silybin besides targeting hepatocytes, and immune cells.[Pubmed: 25149982] | Silymarin, a hepatoprotective agent, has poor oral bioavailability. However, the current dosage form of the drug does not target the liver and inflammatory cells selectively. The aim of the present study was to develop lecithin-based carrier system of Silymarin by incorporating phytosomal-liposomal approach to increase its oral bioavailability and to make it target-specific to the liver for enhanced hepatoprotection.
METHODS AND RESULTS:
The formulation was prepared by film hydration method. Release of drug was assessed at pH 1.2 and 7.4. Formulation was assessed for in vitro hepatoprotection on Chang liver cells, lipopolysaccharide-induced reactive oxygen species (ROS) production by RAW 267.4 (murine macrophages), in vivo efficacy against paracetamol-induced hepatotoxicity and pharmacokinetic study by oral route in Wistar rat.
The formulation showed maximum entrapment (55%) for a lecithin-cholesterol ratio of 6:1. Comparative release profile of formulation was better than Silymarin at pH 1.2 and pH 7.4. In vitro studies showed a better hepatoprotection efficacy for formulation (one and half times) and better prevention of ROS production (ten times) compared to Silymarin. In in vivo model, paracetamol showed significant hepatotoxicity in Wistar rats assessed through LFT, antioxidant markers and inflammatory markers. The formulation was found more efficacious than Silymarin suspension in protecting the liver against paracetamol toxicity and the associated inflammatory conditions. The liposomal formulation yielded a three and half fold higher bioavailability of Silymarin as compared with Silymarin suspension.
CONCLUSIONS:
Incorporating the phytosomal form of Silymarin in liposomal carrier system increased the oral bioavailability and showed better hepatoprotection and better anti-inflammatory effects compared with Silymarin suspension. | | Int J Oncol. 2005 Jan;26(1):169-76. | | Silymarin and skin cancer prevention: anti-inflammatory, antioxidant and immunomodulatory effects (Review).[Pubmed: 15586237] | Several environmental and genetic factors are involved in skin cancer induction, however exposure to chemical carcinogens and solar ultraviolet (UV) radiation are primarily responsible for several skin diseases including skin cancer. Chronic exposure of solar UV radiation to the skin leads to basal cell and squamous cell carcinoma, and melanoma. Chemoprevention of skin cancer by consumption of naturally occurring botanicals appears a practical approach and therefore world-wide interest is considerably increasing to use these botanicals. Sunscreens are useful but their protection is not ideal because of inadequate use, incomplete spectral protection and toxicity.
METHODS AND RESULTS:
Silymarin, a plant flavonoid isolated from the seeds of milk thistle (Silybum marianum), has been shown to have chemopreventive effects against chemical carcinogenesis as well as photocarcinogenesis in various animal tumor models. Topical treatment of Silymarin inhibited 7,12-dimethylbenz(a)anthracene-initiated and several tumor promoters, like 12-O-tetradecanoylphorbol-13-acetate, mezerein, benzoyal peroxide and okadaic acid, induced skin carcinogenesis in mouse models. Similarly, Silymarin also prevented UVB-induced skin carcinogenesis. Wide range of in vivo mechanistic studies indicated that Silymarin possesses antioxidant, anti-inflammatory and immunomodulatory properties which may lead to the prevention of skin cancer in in vivo animal models.
CONCLUSIONS:
The available experimental information suggests that Silymarin is a promising chemopreventive and pharmacologically safe agent which can be exploited or tested against skin cancer in human system. Moreover, Silymarin may favorably supplement sunscreen protection and provide additional anti-photocarcinogenic protection. |
|






 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)