| Structure Identification: |
| Chinese Journal of Experimental Traditional Medical Formulae, 2016 (5) :45-53. | | Chemical Components of Alkaloids from Euodiae Fructus and Their Anti-angiogenic Activities[Reference: WebLink] | To study the chemical constituents and their anti-angiogenic activities of the fruits of Evodia rutaecarpa.
METHODS AND RESULTS:
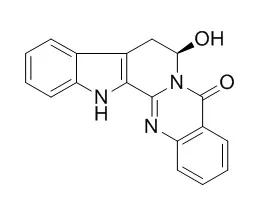
Compounds were isolated and purified by column chromatography using silica gel,Sephadex LH-20, ODS and semi-preparative HPLC. Their structures were identified on the basis of physicochemical properties and spectral data. The anti-angiogenic activities of compounds were evaluated using a zebrafish model. Result: Twenty compounds were isolated and identified from the fruits of E. rutaecarpa,including one degraded limonoids calodendrolide( 1),6 indole alkaloids,i. e. rutaecarpine( 2),evodiamine( 3),goshuyuamide-Ι( 4),N-formyldihydrorutaecarpine( 5),and 7beta-Hydroxyrutaecarpine( 6),11 quinolone alkaloids,i. e. 2-hydroxy-4-methoxy-3-( 3'-methyl-2'-butenyl)-quinoline( 7),1-methyl-2-nonyl-4( 1H)-quinolone( 8),1-methyl-2-decyl-4( 1H)-quinolone( 9),1-methyl-2-undecyl-4( 1H)-quinolone( 10),dihydroevocarpine( 11),1-methyl-2-pentadecenyl-4( 1H)-quinolone( 12),1-methyl-2- [( Z)-6-undecyl]-4( 1H)-quinolone( 13),evocarpine( 14),1-methyl-2- [( Z)-4-tridecyl]-4( 1H)-quinolone( 15),mixture of 1-methyl-2- [( Z)-10-pentadecenyl ]-4( 1H)-quinolone and 1-methyl-2- [( Z)-6-pentadecenyl]-4( 1H)-quinolone( 16),1-methyl-2- [( 6Z,9Z)-6,9-pentadecenyl]-4( 1H)-quinolone( 17),2 amides,i. e. N-methylanthranylamide( 18),acetamide( 19),and one sterol,i. e. β-sitosterol( 20).Compounds 1( 20 mg·L~(-1)),2( 0. 5 mg·L~(-1)),3( 5,10 μg·L~(-1)),4( 10 mg·L~(-1)),5( 50 mg·L~(-1)),10( 20 mg·L~(-1)),11( 50 mg·L~(-1)),16( 20 mg·L~(-1)),and 18( 50 mg·L~(-1)) showed anti-angiogenic effects on internode blood vessels of Zebrafish models.
CONCLUSIONS:
Compounds 1-18 were characteristic constituents of Euodiae Fructus,and compounds 1 and 7 were isolated from this plant for the first time. Compounds 1-5,10,11,16,and 18 showed anti-angiogenic effects on internode blood vessels of Zebrafish models. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)