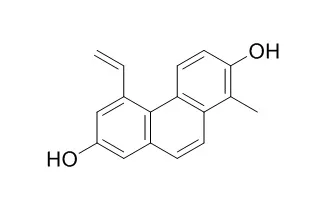
| Description: |
Dehydroeffusol possesses anti-cancer, anxiolytic and sedative properties, it may antagonize the spasmogenic activity of various agents, and therefore, could be a promising agent in the treatment of spasms. Dehydroeffusol displays enhanced antimicrobial activities in light, its antimicrobial activities (minimum inhibitory concentrations) against methicillin-resistant and -sensitive Staphylococcus aureus and Candida albicans are increased 16 fold by irradiation with ultraviolet A (UVA). |
| In vitro: |
| Biochem Pharmacol. 2016 Mar 15;104:8-18. | | Dehydroeffusol inhibits gastric cancer cell growth and tumorigenicity by selectively inducing tumor-suppressive endoplasmic reticulum stress and a moderate apoptosis.[Pubmed: 26774454 ] | Gastric cancer is ranked as the third leading cause of cancer-related death in the world. Although extensive efforts have been made in recent decades to treat gastric cancer with various anticancer drugs, effective anti-gastric cancer therapeutics to cure the disease are still lacking in the clinics. Therefore, potent novel anti-gastric cancer drugs are greatly needed.
METHODS AND RESULTS:
In this study, we explored a novel anti-gastric cancer agent from a medicinal herb named Juncus effusus and found that the active component Dehydroeffusol (DHE), a small molecular phenanthrene, effectively inhibited gastric cancer cell proliferation and tumorigenesis by inducing tumor suppressive endoplasmic reticulum (ER) stress and by triggering moderate apoptosis. Mechanistic studies revealed that DHE selectively activated the intracellular tumor suppressive stress response by promoting the overexpression of the key ER stress marker DNA damage-inducible transcript 3 (DDIT3), through upregulation of activating transcription factor 4 (ATF4). Concurrently, DHE suppressed the expression of the cell survival and ER stress marker glucose regulated protein of molecular mass 78 (GRP78) via downregulation of the transcription factor ATF6. In addition, DHE markedly activated the stress response signaling pathway MEKK4-MKK3/6-p38-DDIT3, but significantly inhibited ERK signaling.
CONCLUSIONS:
Our data suggest that DHE inhibits gastric cancer cell growth and tumorigenicity through selectively inducing a robust tumor suppressive ER stress response and a moderate apoptosis response. Therefore, DHE may provide a novel drug candidate for further development of potential anti-gastric cancer therapeutics. | | Toxicol Appl Pharmacol. 2015 Sep 1;287(2):98-110. | | Dehydroeffusol effectively inhibits human gastric cancer cell-mediated vasculogenic mimicry with low toxicity.[Pubmed: 25982451] | Accumulated data has shown that various vasculogenic tumor cells, including gastric cancer cells, are able to directly form tumor blood vessels via vasculogenic mimicry, supplying oxygen and nutrients to tumors, and facilitating progression and metastasis of malignant tumors. Therefore, tumor vasculogenic mimicry is a rational target for developing novel anticancer therapeutics. However, effective antitumor vasculogenic mimicry-targeting drugs are not clinically available.
METHODS AND RESULTS:
In this study, we purified 2,7-dihydroxyl-1-methyl-5-vinyl-phenanthrene, termed Dehydroeffusol, from the traditional Chinese medicinal herb Juncus effusus L., and found that Dehydroeffusol effectively inhibited gastric cancer cell-mediated vasculogenic mimicry in vitro and in vivo with very low toxicity. Dehydroeffusol significantly suppressed gastric cancer cell adhesion, migration, and invasion. Molecular mechanistic studies revealed that Dehydroeffusol markedly inhibited the expression of a vasculogenic mimicry master gene VE-cadherin and reduced adherent protein exposure on the cell surface by inhibiting gene promoter activity. In addition, Dehydroeffusol significantly decreased the expression of a key vasculogenic gene matrix metalloproteinase 2 (MMP2) in gastric cancer cells, and diminished MMP2 protease activity.
CONCLUSIONS:
Together, our results showed that Dehydroeffusol effectively inhibited gastric cancer cell-mediated vasculogenic mimicry with very low toxicity, suggesting that Dehydroeffusol is a potential drug candidate for anti-gastric cancer neovascularization and anti-gastric cancer therapy. | | Planta Med. 2014 Aug;80(12):978-83. | | Effects of dehydroeffusol on spasmogen-induced contractile responses of rat intestinal smooth muscles.[Pubmed: 25089735] | Dehydroeffusol is a naturally occurring phenanthrene isolated from Juncus effusus.
METHODS AND RESULTS:
In the context of screening new drugs against gastrointestinal spasms, we investigated its effects on isolated rat jejunum in vitro. Dehydroeffusol (30-90 µM) slightly and transiently enhanced contractions in a concentration-dependent manner but significantly inhibited the contractions induced by KCl (100 mM), (±)-Bay-K8644 (5 µM), pilocarpine (90 µM), and histamine (100 µM).
CONCLUSIONS:
These results show that Dehydroeffusol may antagonize the spasmogenic activity of various agents, and therefore, could be a promising agent in the treatment of spasms. Its potential spasmolytic mechanism is also discussed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)